Consider Again the Decomposition of No2 Discussed Above
Affiliate 12. Kinetics
12.4 Integrated Charge per unit Laws
Learning Objectives
By the end of this department, you volition be able to:
- Explicate the course and function of an integrated rate law
- Perform integrated charge per unit law calculations for zero-, starting time-, and second-social club reactions
- Ascertain half-life and carry out related calculations
- Identify the gild of a reaction from concentration/fourth dimension data
The rate laws we have seen thus far relate the rate and the concentrations of reactants. We tin also decide a 2d grade of each rate constabulary that relates the concentrations of reactants and fourth dimension. These are chosen integrated charge per unit laws. We tin can utilize an integrated rate police to make up one's mind the amount of reactant or product present afterwards a menstruation of fourth dimension or to estimate the time required for a reaction to proceed to a sure extent. For instance, an integrated charge per unit law is used to determine the length of fourth dimension a radioactive cloth must exist stored for its radioactivity to disuse to a safe level.
Using calculus, the differential rate constabulary for a chemical reaction can be integrated with respect to time to give an equation that relates the corporeality of reactant or product nowadays in a reaction mixture to the elapsed fourth dimension of the reaction. This process tin either exist very straightforward or very complex, depending on the complexity of the differential rate police force. For purposes of discussion, we will focus on the resulting integrated rate laws for first-, second-, and zero-order reactions.
Showtime-Order Reactions
An equation relating the rate constant chiliad to the initial concentration [A]0 and the concentration [A] t present after any given time t can be derived for a first-order reaction and shown to exist:
[latex]\text{ln}\;(\frac{[A]_t}{[A]_0}) = -kt[/latex]
or
[latex]\text{ln}\;(\frac{[A]_0}{[A]_t}) = kt[/latex]
or
[latex][A] = [A]_0e^{-kt}[/latex]
Example one
The Integrated Rate Law for a Starting time-Order Reaction
The charge per unit constant for the first-order decomposition of cyclobutane, CfourHeight at 500 °C is nine.ii × x−3 south−1:
[latex]\text{C}_4\text{H}_8\;{\longrightarrow}\;2\text{C}_2\text{H}_4[/latex]
How long will it have for eighty.0% of a sample of C4H8 to decompose?
Solution
We use the integrated form of the charge per unit law to answer questions regarding time:
[latex]\text{ln}\;(\frac{[A]_0}{[A]}) = kt[/latex]
There are four variables in the rate police force, and then if we know three of them, we can decide the fourth. In this case we know [A]0, [A], and k, and need to notice t.
The initial concentration of C4H8, [A]0, is not provided, but the provision that 80.0% of the sample has decomposed is enough information to solve this problem. Let x be the initial concentration, in which case the concentration after 80.0% decomposition is 20.0% of x or 0.200x. Rearranging the rate constabulary to isolate t and substituting the provided quantities yields:
[latex]\begin{assortment}{r @{{}={}} l} t & \text{ln}\;\frac{[x]}{[0.200x]}\;\times\;\frac{ane}{1000} \\[0.5em] & \text{ln}\;\frac{0.100\;\text{mol\;L}^{-1}}{0.020\;\text{mol\;L}^{-1}}\;\times\;\frac{1}{9.two\;\times\;x^{-3}\text{south}^{-one}} \\[0.5em] & ane.609\;\times\;\frac{one}{ix.ii\;\times\;10^{-3}\text{due south}^{-ane}} \\[0.5em] & 1.7\;\times\;x^2\text{due south} \terminate{array}[/latex]
Bank check Your Learning
Iodine-131 is a radioactive isotope that is used to diagnose and treat some forms of thyroid cancer. Iodine-131 decays to xenon-131 according to the equation:
[latex]\text{I-}131\;{\longrightarrow}\;\text{Xe-}131\;+\;\text{electron}[/latex]
The disuse is outset-guild with a rate constant of 0.138 d−one. All radioactive decay is first order. How many days will information technology take for 90% of the iodine−131 in a 0.500 M solution of this substance to decay to Xe-131?
Nosotros can use integrated rate laws with experimental data that consist of time and concentration information to decide the order and rate constant of a reaction. The integrated charge per unit police can be rearranged to a standard linear equation format:
[latex]\begin{assortment}{r @{{}={}} 50} \text{ln}[A] & (-k)(t)\;+\;\text{ln}[A]_0 \\[0.5em] y & mx\;+\;b \end{assortment}[/latex]
A plot of ln[A] versus t for a first-order reaction is a straight line with a slope of −1000 and an intercept of ln[A]0. If a set of rate data are plotted in this way but do not result in a direct line, the reaction is not first order in A.
Example 2
Determination of Reaction Order by Graphing
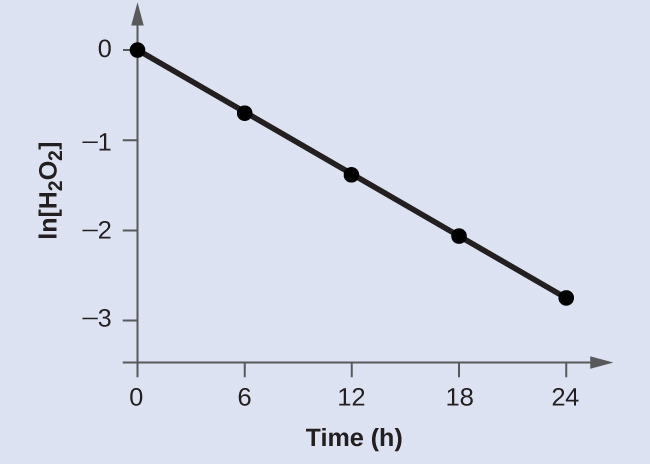
Bear witness that the data in Effigy i in Chapter 12.one Chemical Reaction Rates tin be represented by a first-order charge per unit law by graphing ln[HtwoOtwo] versus time. Decide the charge per unit constant for the rate of decomposition of H2O2 from this data.
Solution
The data from Figure 1 in Chapter 12.1 Chemic Reaction Rates with the addition of values of ln[H2Oii] are given in Figure 1.
| Trial | Time (h) | [H2O2] (G) | ln[H2Otwo] |
|---|---|---|---|
| 1 | 0 | 1.000 | 0.0 |
| 2 | 6.00 | 0.500 | −0.693 |
| iii | 12.00 | 0.250 | −ane.386 |
| 4 | eighteen.00 | 0.125 | −2.079 |
| v | 24.00 | 0.0625 | −two.772 |
| Table 17. | |||
The plot of ln[HtwoO2] versus fourth dimension is linear, thus we take verified that the reaction may be described by a kickoff-order rate law.
The rate abiding for a showtime-lodge reaction is equal to the negative of the slope of the plot of ln[H2O2] versus fourth dimension where:
[latex]\text{slope} = \frac{\text{change\;in}\;y}{\text{modify\;in}\;x} = \frac{{\Delta}y}{{\Delta}ten} = \frac{{\Delta}\text{ln}[\text{H}_2\text{O}_2]}{{\Delta}t}[/latex]
In gild to decide the slope of the line, nosotros need two values of ln[HtwoO2] at different values of t (i near each end of the line is preferable). For example, the value of ln[HtwoO2] when t is six.00 h is −0.693; the value when t = 12.00 h is −1.386:
[latex]\brainstorm{array}{r @{{}={}} l} \text{slope} & \frac{-1.386\;-\;(-0.693)}{12.00\;\text{h}\;-\;6.00\;\text{h}} \\[0.5em] & \frac{-0.693}{6.00\;\text{h}} \\[0.5em] & -i.155\;\times\;10^{-one}\text{h}^{-1} \\[0.5em] k & -\text{slope} = -(-1.155\;\times\;10^{-1}\text{h}^{-1}) = i.155\;\times\;ten^{-1}\text{h}^{-1} \end{array}[/latex]
Bank check Your Learning
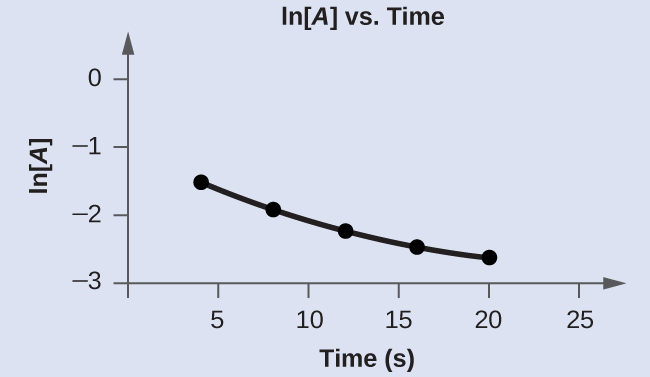
Graph the post-obit data to decide whether the reaction [latex]A\;{\longrightarrow}\;B\;+\;C[/latex] is first club.
| Trial | Time (s) | [A] |
|---|---|---|
| 1 | 4.0 | 0.220 |
| two | 8.0 | 0.144 |
| 3 | 12.0 | 0.110 |
| four | xvi.0 | 0.088 |
| 5 | 20.0 | 0.074 |
| Tabular array 18. | ||
Answer:
The plot of ln[A] vs. t is not a direct line. The equation is not first order:
2nd-Order Reactions
The equations that relate the concentrations of reactants and the rate constant of 2d-club reactions are fairly complicated. We volition limit ourselves to the simplest second-social club reactions, namely, those with rates that are dependent upon just ane reactant's concentration and described by the differential rate law:
[latex]\text{Charge per unit} = k[A]^2[/latex]
For these second-order reactions, the integrated rate law is:
[latex]\frac{1}{[A]} = kt\;+\;\frac{ane}{[A]_0}[/latex]
where the terms in the equation have their usual meanings as divers earlier.
Example iii
The Integrated Rate Law for a Second-Order Reaction
The reaction of butadiene gas (CfourHvi) with itself produces C8H12 gas equally follows:
[latex]2\text{C}_4\text{H}_6(g)\;{\longrightarrow}\;\text{C}_8\text{H}_{12}(thou)[/latex]
The reaction is second order with a charge per unit constant equal to 5.76 × ten−2 L/mol/min under certain weather. If the initial concentration of butadiene is 0.200 M, what is the concentration remaining later on ten.0 min?
Solution
We use the integrated form of the rate police force to answer questions regarding time. For a second-gild reaction, we have:
[latex]\frac{1}{[A]} = kt\;+\;\frac{ane}{[A]_0}[/latex]
We know three variables in this equation: [A]0 = 0.200 mol/L, k = five.76 × 10−2 L/mol/min, and t = 10.0 min. Therefore, nosotros can solve for [A], the fourth variable:
[latex]\begin{array}{r @{{}={}} 50} \frac{i}{[A]} & (5.76\;\times\;10^{-two}\text{L\;mol}^{-1}\text{min}^{-1})(10\;\text{min})\;+\;\frac{one}{0.200\;\text{mol}^{-1}} \\[0.5em] \frac{i}{[A]} & (5.76\;\times\;x^{-one}\text{L\;mol}^{-1})\;+\;5.00\;\text{L\;mol}^{-one} \\[0.5em] \frac{ane}{[A]} & 5.58\;\text{L\;mol}^{-1} \\[0.5em] [A] & one.79\;\times\;10^{-1}\;\text{mol\;L}^{-one} \end{array}[/latex]
Therefore 0.179 mol/L of butadiene remain at the end of 10.0 min, compared to the 0.200 mol/50 that was originally present.
Check Your Learning
If the initial concentration of butadiene is 0.0200 M, what is the concentration remaining after 20.0 min?
The integrated rate police force for our 2nd-order reactions has the form of the equation of a straight line:
[latex]\begin{assortment}{r @{{}={}} 50} \frac{1}{[A]} & kt\;+\;\frac{1}{[A]_0} \\[0.5em] y & mx\;+\;b \end{assortment}[/latex]
A plot of [latex]\frac{1}{[A]}[/latex] versus t for a second-social club reaction is a directly line with a slope of k and an intercept of [latex]\frac{1}{[A]_0}[/latex]. If the plot is not a straight line, and then the reaction is not 2nd order.
Example 4
Determination of Reaction Gild past Graphing
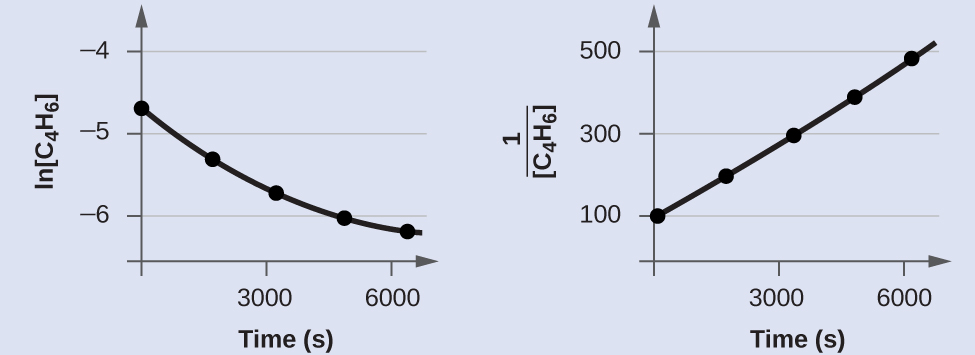
Exam the data given to prove whether the dimerization of C4H6 is a showtime- or a second-guild reaction.
Solution
| Trial | Fourth dimension (s) | [CfourHvi] (M) |
|---|---|---|
| 1 | 0 | 1.00 × 10−ii |
| two | 1600 | 5.04 × 10−3 |
| 3 | 3200 | 3.37 × 10−3 |
| iv | 4800 | ii.53 × 10−3 |
| five | 6200 | ii.08 × 10−three |
| Table 19. | ||
In gild to distinguish a first-order reaction from a second-gild reaction, we plot ln[C4Hhalf-dozen] versus t and compare information technology with a plot of [latex]\frac{1}{[\text{C}_4\text{H}_6]}[/latex] versus t. The values needed for these plots follow.
| Time (due south) | [latex]\frac{one}{[\text{C}_4\text{H}_6]}(M^{-1})[/latex] | ln[C4Hsix] |
|---|---|---|
| 0 | 100 | −4.605 |
| 1600 | 198 | −5.289 |
| 3200 | 296 | −five.692 |
| 4800 | 395 | −5.978 |
| 6200 | 481 | −half dozen.175 |
| Table 20. | ||
The plots are shown in Effigy 2. As you can meet, the plot of ln[C4H6] versus t is not linear, therefore the reaction is not first order. The plot of [latex]\frac{1}{[\text{C}_4\text{H}_6]}[/latex] versus t is linear, indicating that the reaction is second order.
Check Your Learning
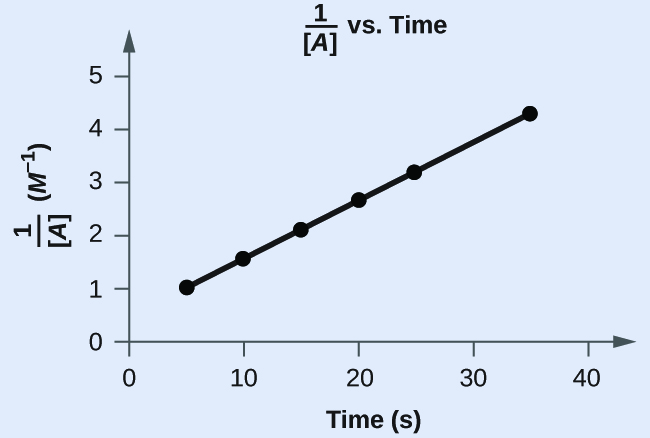
Does the following information fit a 2d-social club rate constabulary?
| Trial | Fourth dimension (due south) | [A] (One thousand) |
|---|---|---|
| one | 5 | 0.952 |
| 2 | 10 | 0.625 |
| 3 | 15 | 0.465 |
| four | twenty | 0.370 |
| 5 | 25 | 0.308 |
| half-dozen | 35 | 0.230 |
| Tabular array 21. | ||
Answer:
Yes. The plot of [latex]\frac{1}{[A]}[/latex] vs. t is linear:
Zero-Order Reactions
For zero-order reactions, the differential rate police force is:
[latex]\text{Rate} = yard[A]^0 = one thousand[/latex]
A naught-lodge reaction thus exhibits a constant reaction rate, regardless of the concentration of its reactants.
The integrated rate police force for a cipher-order reaction also has the form of the equation of a straight line:
[latex]\begin{assortment}{r @{{}={}} l} [A] & -kt\;+\;[A]_0 \\[0.5em] y & mx\;+\;b \end{assortment}[/latex]
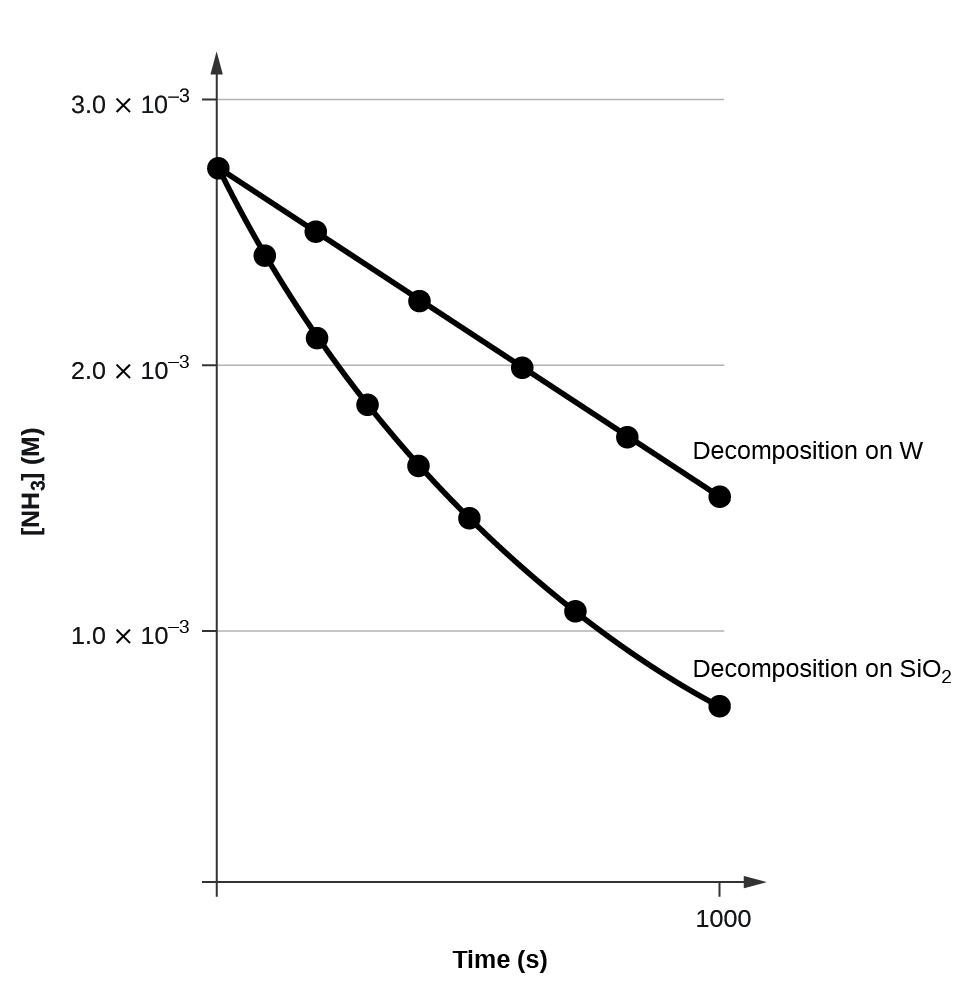
A plot of [A] versus t for a zilch-society reaction is a directly line with a slope of −k and an intercept of [A]0. Effigy 3 shows a plot of [NH3] versus t for the decomposition of ammonia on a hot tungsten wire and for the decomposition of ammonia on hot quartz (SiOii). The decomposition of NHiii on hot tungsten is zero guild; the plot is a straight line. The decomposition of NH3 on hot quartz is non zero order (it is first social club). From the slope of the line for the null-lodge decomposition, we tin can determine the charge per unit abiding:
[latex]\text{slope} = -k = i.3110^{-vi}\;\text{mol}/\text{L}/\text{s}[/latex]
The Half-Life of a Reaction
The half-life of a reaction (t one/2) is the fourth dimension required for one-half of a given amount of reactant to be consumed. In each succeeding half-life, one-half of the remaining concentration of the reactant is consumed. Using the decomposition of hydrogen peroxide (Figure 1 in Chapter 12.1 Chemical Reaction Rates) as an instance, we find that during the start half-life (from 0.00 hours to 6.00 hours), the concentration of HtwoO2 decreases from 1.000 Thousand to 0.500 Yard. During the second half-life (from half dozen.00 hours to 12.00 hours), it decreases from 0.500 M to 0.250 M; during the third one-half-life, information technology decreases from 0.250 M to 0.125 Thou. The concentration of HiiOii decreases past half during each successive period of half-dozen.00 hours. The decomposition of hydrogen peroxide is a first-order reaction, and, as can exist shown, the half-life of a showtime-order reaction is independent of the concentration of the reactant. Still, half-lives of reactions with other orders depend on the concentrations of the reactants.
First-Society Reactions
We can derive an equation for determining the one-half-life of a first-guild reaction from the alternate course of the integrated rate law equally follows:
[latex]\begin{assortment}{r @{{}={}} l} \text{ln}\;\frac{[A]_0}{[A]} & kt \\[0.5em] t & \text{ln}\;\frac{[A]_0}{[A]}\;\times\;\frac{1}{thou} \end{assortment}[/latex]
If we set up the time t equal to the half-life, [latex]t_{1/2}[/latex], the respective concentration of A at this time is equal to one-half of its initial concentration. Hence, when [latex]t = t_{1/2}[/latex], [latex][A] = \frac{1}{two}[A]_0[/latex].
Therefore:
[latex]\begin{array}{r @{{}={}} l} t_{ane/2} & \text{ln}\;\frac{[A]_0}{\frac{ane}{ii}[A]_0}\;\times\;\frac{one}{k} \\[0.5em] & \text{ln}\;2\;\times\;\frac{i}{k} = 0.693\;\times\;\frac{ane}{yard} \end{assortment}[/latex]
Thus:
[latex]t_{1/2} = \frac{0.693}{k}[/latex]
Nosotros tin come across that the half-life of a starting time-society reaction is inversely proportional to the charge per unit constant k. A fast reaction (shorter half-life) will have a larger g; a deadening reaction (longer half-life) will have a smaller k.
Instance five
Calculation of a Start-social club Rate Constant using One-half-Life
Calculate the rate constant for the first-order decomposition of hydrogen peroxide in water at forty °C, using the information given in Figure 4.

Solution
The one-half-life for the decomposition of HtwoO2 is ii.sixteen × 104 south:
[latex]\begin{array}{r @{{}={}} l} t_{1/two} & \frac{0.693}{g} \\[0.5em] thousand & \frac{0.693}{t_{1/ii}} = \frac{0.693}{2.xvi\;\times\;x^iv\text{s}} = 3.21\;\times\;10^{-5}\text{southward}^{-1} \end{array}[/latex]
Check Your Learning
The kickoff-lodge radioactive decay of iodine-131 exhibits a charge per unit constant of 0.138 d−one. What is the half-life for this decay?
Second-Order Reactions
Nosotros can derive the equation for calculating the one-half-life of a second order equally follows:
[latex]\frac{1}{[A]} = kt\;+\;\frac{ane}{[A]_0}[/latex]
or
[latex]\frac{1}{[A]}\;-\;\frac{1}{[A]_0} = kt[/latex]
If
[latex]t = t_{ane/2}[/latex]
then
[latex][A] = \frac{1}{2}[A]_0[/latex]
and nosotros can write:
[latex]\begin{array}{r @{{}={}} l} \frac{1}{\frac{ane}{2}[A]_0}\;-\;\frac{one}{[A]_0} & kt_{1/2} \\[0.5em] ii[A]_0\;-\;\frac{1}{[A]_0} & kt_{1/ii} \\[0.5em] \frac{1}{[A]_0} & kt_{1/2} \finish{array}[/latex]
Thus:
[latex]t_{1/2} = \frac{1}{g[A]_0}[/latex]
For a second-order reaction, [latex]t_{1/2}[/latex] is inversely proportional to the concentration of the reactant, and the half-life increases equally the reaction proceeds because the concentration of reactant decreases. Consequently, we observe the apply of the one-half-life concept to exist more than complex for second-lodge reactions than for commencement-social club reactions. Unlike with first-gild reactions, the rate constant of a second-order reaction cannot be calculated directly from the half-life unless the initial concentration is known.
Nothing-Order Reactions
Nosotros tin derive an equation for calculating the half-life of a zero gild reaction as follows:
[latex][A] = -kt\;+\;[A]_0[/latex]
When half of the initial amount of reactant has been consumed [latex]t = t_{i/2}[/latex] and [latex][A] = \frac{[A]_0}{2}[/latex]. Thus:
[latex]\begin{array}{r @{{}={}} l} \frac{[A]_0}{2} & -kt_{one/2}\;+\;[A]_0 \\[0.5em] kt_{i/two} & \frac{[A]_0}{two} \end{array}[/latex]
and
[latex]t_{ane/2} = \frac{[A]_0}{2k}[/latex]
The half-life of a zero-order reaction increases as the initial concentration increases.
Equations for both differential and integrated rate laws and the corresponding one-half-lives for zero-, first-, and second-order reactions are summarized in Tabular array 22.
| Zero-Order | First-Order | 2d-Order | |
|---|---|---|---|
| rate constabulary | rate = k | rate = k[A] | rate = thou[A]2 |
| units of charge per unit constant | M s−1 | s−1 | M −1 s−1 |
| integrated charge per unit law | [A] = −kt + [A]0 | ln[A] = −kt + ln[A]0 | [latex]\frac{one}{[A]} = kt\;+\;(\frac{1}{[A]_0})[/latex] |
| plot needed for linear fit of rate data | [A] vs. t | ln[A] vs. t | [latex]\frac{i}{[A]}\;\text{vs.}\;t[/latex] |
| human relationship between slope of linear plot and rate abiding | k = −slope | 1000 = −slope | g = +slope |
| one-half-life | [latex]t_{one/ii} = \frac{[A]_0}{2k}[/latex] | [latex]t_{one/2} = \frac{0.693}{k}[/latex] | [latex]t_{ane/two} = \frac{1}{[A]_0k}[/latex] |
| Table 22. Summary of Rate Laws for Null-, First-, and Second-Order Reactions | |||
Central Concepts and Summary
Differential rate laws can be adamant past the method of initial rates or other methods. We measure values for the initial rates of a reaction at different concentrations of the reactants. From these measurements, we determine the order of the reaction in each reactant. Integrated rate laws are determined by integration of the respective differential rate laws. Rate constants for those rate laws are determined from measurements of concentration at diverse times during a reaction.
The half-life of a reaction is the time required to decrease the corporeality of a given reactant by one-half. The half-life of a nothing-lodge reaction decreases equally the initial concentration of the reactant in the reaction decreases. The one-half-life of a first-order reaction is contained of concentration, and the half-life of a second-club reaction decreases as the concentration increases.
Central Equations
- integrated rate law for zero-society reactions: [latex][A] = -kt\;+\;[A]_0[/latex], [latex]t_{ane/2} = \frac{[A]_0}{2k}[/latex]
- integrated rate law for outset-order reactions: [latex]\text{ln}\;[A] = -kt\;+\;\text{ln}\;[A]_0[/latex], [latex]t_{1/ii} = \frac{0.693}{thousand}[/latex]
- integrated charge per unit police force for second-gild reactions: [latex]\frac{ane}{[A]} = kt\;+\;\frac{1}{[A]_0}[/latex], [latex]t_{1/2} = \frac{1}{[A]_0k}[/latex]
Chemistry End of Chapter Exercises
- Draw how graphical methods tin can exist used to determine the society of a reaction and its rate constant from a serial of data that includes the concentration of A at varying times.
- Utilize the data provided to graphically determine the lodge and charge per unit constant of the post-obit reaction: [latex]\text{SO}_2\text{Cl}_2\;{\longrightarrow}\;\text{Then}_2\;+\;\text{Cl}_2[/latex]
Time (due south) 0 5.00 × 103 1.00 × ten4 one.50 × 10four [And so2Cltwo] (Yard) 0.100 0.0896 0.0802 0.0719 Time (s) 2.fifty × x4 3.00 × 10four 4.00 × tenfour [And then2Cl2] (M) 0.0577 0.0517 0.0415 Table 23. - Employ the information provided in a graphical method to determine the social club and rate constant of the following reaction:
[latex]2P\;{\longrightarrow}\;Q\;+\;West[/latex]Time (s) 9.0 xiii.0 18.0 22.0 25.0 [P] (One thousand) 1.077 × x−3 1.068 × ten−3 ane.055 × 10−3 ane.046 × 10−three i.039 × 10−3 Tabular array 24. - Pure ozone decomposes slowly to oxygen, [latex]2\text{O}_3(chiliad)\;{\longrightarrow}\;three\text{O}_2(thousand)[/latex]. Use the data provided in a graphical method and determine the gild and rate constant of the reaction.
Time (h) 0 ii.0 × 103 seven.6 × 103 1.00 × 104 [O3] (M) 1.00 × ten−5 iv.98 × 10−6 2.07 × ten−6 i.66 × 10−6 Fourth dimension (h) i.23 × x4 1.43 × 104 1.lxx × 10iv [Othree] (K) i.39 × 10−6 1.22 × ten−six 1.05 × 10−six Table 25. - From the given data, use a graphical method to decide the order and rate constant of the post-obit reaction:
[latex]2X\;{\longrightarrow}\;Y\;+\;Z[/latex]Time (s) 5.0 ten.0 15.0 twenty.0 25.0 30.0 35.0 forty.0 [X] (M) 0.0990 0.0497 0.0332 0.0249 0.0200 0.0166 0.0143 0.0125 Table 26. - What is the one-half-life for the kickoff-guild decay of phosphorus-32? ([latex]_{fifteen}^{32}\text{P}\;{\longrightarrow}\;_{xvi}^{32}\text{S}\;+\;\text{eastward}^{-})[/latex]. The charge per unit constant for the decay is 4.85 × 10−ii day−1.
- What is the one-half-life for the first-social club disuse of carbon-14? [latex](_6^{14}\text{C}\;{\longrightarrow}\;_7^{xiv}\text{N}\;+\;\text{due east}^{-})[/latex] The rate constant for the decay is 1.21 × 10−iv twelvemonth−one.
- What is the half-life for the decomposition of NOCl when the concentration of NOCl is 0.fifteen M? The rate constant for this second-society reaction is eight.0 × 10−8 L/mol/southward.
- What is the one-half-life for the decomposition of Othree when the concentration of O3 is 2.35 × x−6 Thou? The rate constant for this 2nd-club reaction is l.4 Fifty/mol/h.
- The reaction of compoundA to give compoundsC andD was found to exist 2d-society inA. The charge per unit constant for the reaction was adamant to be two.42 L/mol/s. If the initial concentration is 0.500 mol/Fifty, what is the value of tone/2?
- The half-life of a reaction of compound A to requite compounds D and East is 8.fifty min when the initial concentration of A is 0.150 mol/L. How long will it take for the concentration to drop to 0.0300 mol/L if the reaction is (a) first order with respect to A or (b) second society with respect to A?
- Some leaner are resistant to the antibiotic penicillin because they produce penicillinase, an enzyme with a molecular weight of 3 × 104 g/mol that converts penicillin into inactive molecules. Although the kinetics of enzyme-catalyzed reactions tin can be complex, at depression concentrations this reaction can be described past a rate equation that is get-go order in the catalyst (penicillinase) and that also involves the concentration of penicillin. From the following data: 1.0 L of a solution containing 0.fifteen µg (0.fifteen × 10−6 g) of penicillinase, determine the order of the reaction with respect to penicillin and the value of the rate constant.
[Penicillin] (M) Rate (mol/L/min) 2.0 × 10−6 i.0 × ten−ten 3.0 × ten−six one.5 × x−10 4.0 × x−vi 2.0 × ten−10 Tabular array 27. - Both technetium-99 and thallium-201 are used to image middle muscle in patients with suspected heart bug. The one-half-lives are six h and 73 h, respectively. What percent of the radioactivity would remain for each of the isotopes after 2 days (48 h)?
- There are 2 molecules with the formula CthreeH6. Propene, [latex]\text{CH}_3\text{CH}=\text{CH}_2[/latex], is the monomer of the polymer polypropylene, which is used for indoor-outdoor carpets. Cyclopropane is used every bit an coldhearted:
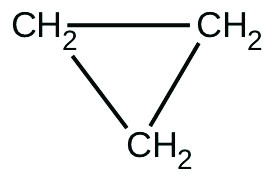
When heated to 499 °C, cyclopropane rearranges (isomerizes) and forms propene with a rate constant of
5.95 × x−4 s−1. What is the half-life of this reaction? What fraction of the cyclopropane remains afterwards 0.75 h at 499.v °C?
- Fluorine-18 is a radioactive isotope that decays by positron emission to form oxygen-18 with a half-life of 109.7 min. (A positron is a particle with the mass of an electron and a unmarried unit of measurement of positive charge; the equation is [latex]_{518}^{9}\text{F}\;{\longrightarrow}\;_{18}^{eight}\text{O}\;+\;\text{e}^{-}[/latex].) Physicians use 18F to study the brain by injecting a quantity of fluoro-substituted glucose into the blood of a patient. The glucose accumulates in the regions where the brain is active and needs nourishment.
(a) What is the charge per unit abiding for the decomposition of fluorine-18?
(b) If a sample of glucose containing radioactive fluorine-xviii is injected into the blood, what percent of the radioactivity will remain later v.59 h?
(c) How long does it have for 99.99% of the 18F to disuse?
- Suppose that the one-half-life of steroids taken by an athlete is 42 days. Assuming that the steroids biodegrade by a outset-gild process, how long would information technology take for [latex]\frac{one}{64}[/latex] of the initial dose to remain in the athlete's trunk?
- Recently, the skeleton of King Richard III was found under a parking lot in England. If tissue samples from the skeleton contain about 93.79% of the carbon-xiv expected in living tissue, what year did King Richard III die? The one-half-life for carbon-14 is 5730 years.
- Nitroglycerine is an extremely sensitive explosive. In a series of advisedly controlled experiments, samples of the explosive were heated to 160 °C and their commencement-order decomposition studied. Decide the average rate constants for each experiment using the following data:
Initial [C3HvDue northiiiO9] (M) 4.88 3.52 2.29 one.81 5.33 4.05 2.95 one.72 t (s) 300 300 300 300 180 180 180 180 % Decomposed 52.0 52.9 53.2 53.9 34.six 35.9 36.0 35.four Table 28. - For the past 10 years, the unsaturated hydrocarbon 1,3-butadiene ([latex]\text{CH}_2=\text{CH}-\text{CH}=\text{CH}_2[/latex]) has ranked 38th amongst the top 50 industrial chemicals. It is used primarily for the manufacture of constructed rubber. An isomer exists besides equally cyclobutene:
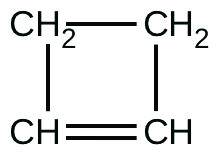
The isomerization of cyclobutene to butadiene is outset-society and the rate constant has been measured as 2.0 × x−4 southward−1 at 150 °C in a 0.53-L flask. Decide the partial pressure of cyclobutene and its concentration after 30.0 minutes if an isomerization reaction is carried out at 150 °C with an initial force per unit area of 55 torr.
Glossary
- half-life of a reaction (t l/ii)
- time required for half of a given amount of reactant to be consumed
- integrated rate law
- equation that relates the concentration of a reactant to elapsed fourth dimension of reaction
Solutions
Answers to Chemistry Terminate of Chapter Exercises
ii. Plotting a graph of ln[Then2Cl2] versus t reveals a linear trend; therefore we know this is a first-order reaction:
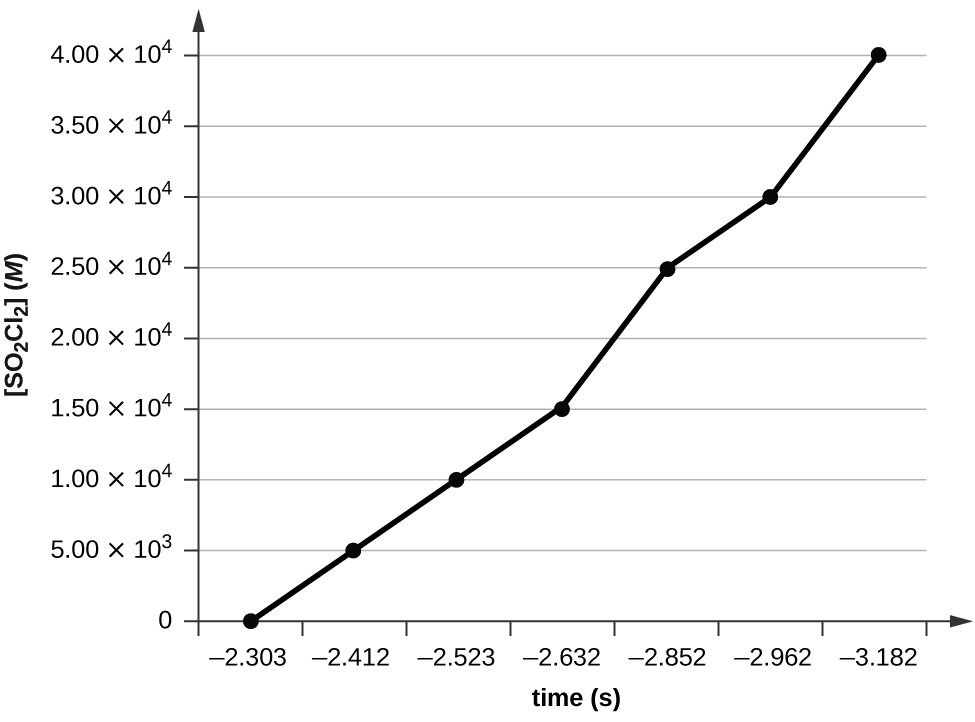
k = −ii.20 × 10five s−1
4.
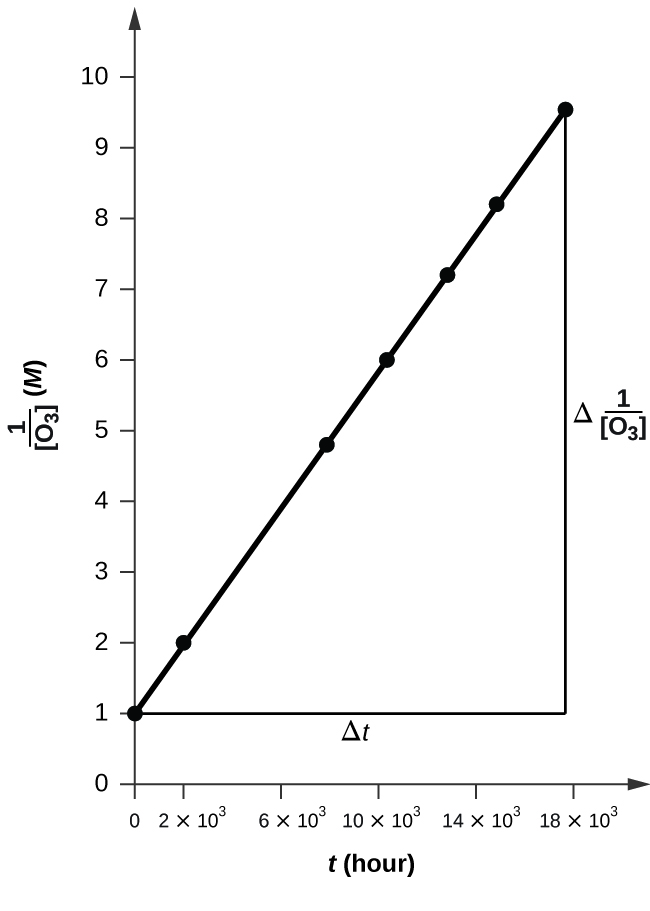
The plot is nicely linear, and so the reaction is second order.
k = 50.1 L mol−one h−1
6. 14.three d
8. eight.3 × 107 south
10. 0.826 southward
12. The reaction is first order.
thousand = i.0 × 10seven mol−ane min−i
14. 4.98; xx% remains
16. 252 days
18.
| [A]0 (Thou) | one thousand × 10iii (s−1) |
|---|---|
| four.88 | ii.45 |
| 3.52 | 2.51 |
| ii.29 | 2.54 |
| 1.81 | 2.58 |
| 5.33 | 2.35 |
| iv.05 | 2.44 |
| 2.95 | two.47 |
| 1.72 | 2.43 |
| Table 29. | |
Source: https://opentextbc.ca/chemistry/chapter/12-4-integrated-rate-laws/
0 Response to "Consider Again the Decomposition of No2 Discussed Above"
Post a Comment